Enzyme Properties
5' to 3' exonuclease activity: No
3' to 5' exonuclease activity: No
3’ – A Overhang: Yes
Strand Displacement: Yes
Fragment Size: Up to 9 kb
Application
First-strand synthesis of cDNA from RNA molecules
RT-PCR
Random priming reaction
Library construction
Probe labeling
mRNA 5’ end mapping by primer extension analysis
Reagents Supplied
5 x Reaction Buffer: 50 mM Tris-HCl, 250 mM KCl, 10 mM MgCl2, pH 8.1
100 mM DTT
dNTP Mix: 2.5 mM of each dNTP
Concentration
200 U/μl
Storage Conditions
50% glycerol containing 20 mM Tris-Cl (pH7.6), 150 mM NaCl, 0.1 mM EDTA, 1 mM DTT, 0.1 % IGEPAL CA-630
Store Temperature
-20°C
Unit Definition
One unit is defined as the amount of enzyme required to incorporates 1 nmole of dTTP into acid-precipitable material in 10 minutes at 37°C using poly (A)ooligo (dT) as template primer.

Figure 1. Comparison of transferrin receptor gene amplification with different reverse transcriptases.
700 ng of total RNA was used for reverse transcription and the same amount of amplified products were used for electrophoresis.
Lane MW: 100 bp Plus DNA Ladder (D-1035)
Lane 1 - 4: TFR (Transferrin receptor gene) amplified with MMLV
Lane 5 - 8: TFR amplified with CycleScript
Lane 9 - 12: TFR amplified with CycleScript
Lane 13 - 16: TFR amplified with MMLV from Company IA
Lane 17 - 20: TFR amplified with S-script from Company S
Lane 21 - 24: TFR amplified with S-script ll from Company I
Lane 25 - 28: TFR amplified with S-script lll from Company I
Lane 29 - 32: TFR amplified with O-script from Company Q
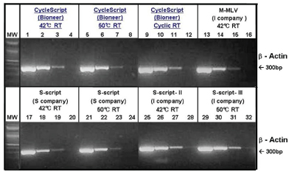
Figure 2. Comparison of β-actin gene amplification with different reverse transcriptases.
Each 10 ng, 1 ng, 100 pg, and 10 pg of total RNA was used for reverse transcription and the same amount of amplified products were used for electrophoresis.
Lane 1 - 4: &beta-actin amplified with CycleScript
Lane 5 - 8: &beta-actin amplified with CycleScript
Lane 9 - 12: &beta-actin amplified with CycleScript
Lane 13 - 16: &beta-actin amplified with MMLV from Company I
Lane 17 - 20: &beta-actin amplified with S-script from Company S
Lane 21 - 24: &beta-actin amplified with S-script from Company S
Lane 25 - 28: &beta-actin amplified with S-script from Company I
Lane 29 - 32: &beta-actin amplified with S-script from Company I
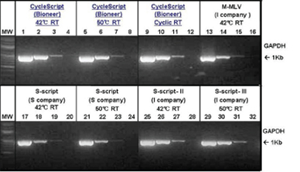
Figure 3. Comparison of GAPDH gene amplification with different reverse transcriptases.
Each 10 ng, 1 ng, 100 pg, and 10 pg of total RNA was used for reverse transcription and the same amount of amplified products were used for electrophoresis.
Lane 1 - 4: GAPDH amplified with CycleScript
Lane 5 - 8: GAPDH amplified with CycleScript
Lane 9 - 12: GAPDH amplified with CycleScript
Lane 13 - 16: GAPDH amplified with MMLV from Company I
Lane 17 - 20: GAPDH amplified with S-script from Company S
Lane 21 - 24: GAPDH amplified with S-script from Company S
Lane 25 - 28: GAPDH amplified with S-script from Company I
Lane 29 - 32: GAPDH amplified with S-script from Company I
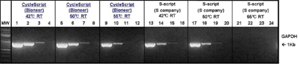
Figure 4. Working temperature comparison of different reverse transcriptases.
Each 10 ng, 1 ng, 100 pg, and 10 pg of total RNA was used for reverse transcription and the same amount of amplified products were used for electrophoresis.
Lane 1 - 4: GAPDH amplified with CycleScript
Lane 5 - 8: GAPDH amplified with CycleScript
Lane 9 - 12: GAPDH amplified with CycleScript
Lane 13 - 16: GAPDH amplified with S-script from company S
Lane 17 - 20: GAPDH amplified with S-script from company S
Lane 21 - 24: GAPDH amplified with S-script from company S
]]>
Manual
• CycleScript Reverse Transcriptase
MSDS
• Cyclescript Rtase
• Cyclescript Rtase-dilution buffer
• Cyclescript Rtase-reaction buffer
Brochure
• Enzymes 2010 Brochure
Quality Assurance
Bioneer is the holder of Quality Management System Certificates for the following standards.
• ISO 9001:2008 - certificate
• EN ISO 13485:2003 / AC:2009 - certificate
• EC Directive 98/79/EC - certificate
]]>